1,2,4-trimethoxybenzene
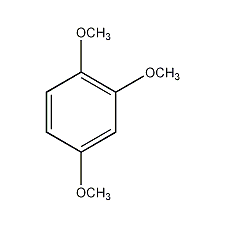

structural formula
| business number | 03qk |
|---|---|
| molecular formula | c9h12o3 |
| molecular weight | 168.19 |
| label |
hydroxyhydroquinone trimethyl ether, aromatic compounds |
numbering system
cas number:135-77-3
mdl number:mfcd00008360
einecs number:205-219-7
rtecs number:none
brn number:2047579
pubchem number:24900410
physical property data
none
toxicological data
none
ecological data
none
molecular structure data
molecular property data:
1、 molar refractive index:46.28
2、 molar volume(m3/mol):161.4
3、 isotonic specific volume(90.2k):377.2
4、 surface tension(3.0 dyne/cm):29.8
5、 polarizability0.5 10-24 cm3):18.35
compute chemical data
1. reference value for hydrophobic parameter calculation (xlogp): none
2. number of hydrogen bond donors: 0
3. number of hydrogen bond acceptors: 3
4. number of rotatable chemical bonds: 3
5. number of tautomers: none
6. topological molecule polar surface area 27.7
7. number of heavy atoms: 12
8. surface charge: 0
9. complexity: 127
10. number of isotope atoms: 0
11. determine the number of atomic stereocenters: 0
12. uncertain number of atomic stereocenters: 0
13. determine the number of chemical bond stereocenters: 0
14. number of uncertain chemical bond stereocenters: 0
15. number of covalent bond units: 1
properties and stability
liquid. boiling point 247℃, relative density 1.106, refractive index 1.5330, flash point >110℃
storage method
none
synthesis method
oxidize vanillin with hydrogen peroxide, then hydrolyze, and methylate it with dimethyl sulfate; or use hydroquinone to oxidize it with sodium dichromate and sulfuric acid to generate terequinone, which is prepared with acetic anhydride. acetylation yields 1,2,4-phloroglucinol triacetate, which is obtained by methylation with dimethyl sulfate.
purpose
intermediates of leucine.
1. reference value for hydrophobic parameter calculation (xlogp): none
2. number of hydrogen bond donors: 0
3. number of hydrogen bond acceptors: 3
4. number of rotatable chemical bonds: 3
5. number of tautomers: none
6. topological molecule polar surface area 27.7
7. number of heavy atoms: 12
8. surface charge: 0
9. complexity: 127
10. number of isotope atoms: 0
11. determine the number of atomic stereocenters: 0
12. uncertain number of atomic stereocenters: 0
13. determine the number of chemical bond stereocenters: 0
14. number of uncertain chemical bond stereocenters: 0
15. number of covalent bond units: 1
properties and stability
liquid. boiling point 247℃, relative density 1.106, refractive index 1.5330, flash point >110℃
storage method
none
synthesis method
oxidize vanillin with hydrogen peroxide, then hydrolyze, and methylate it with dimethyl sulfate; or use hydroquinone to oxidize it with sodium dichromate and sulfuric acid to generate terequinone, which is prepared with acetic anhydride. acetylation yields 1,2,4-phloroglucinol triacetate, which is obtained by methylation with dimethyl sulfate.
purpose
intermediates of leucine.
