2-hydroxyanthraquinone
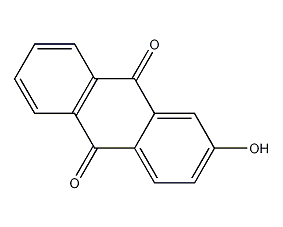

structural formula
| business number | 068j |
|---|---|
| molecular formula | c14h8o3 |
| molecular weight | 224.21 |
| label |
2-hydroxy-9,10-anthracenedione, 2-hydroxy-9,10-anthracenedione |
numbering system
cas number:605-32-3
mdl number:none
einecs number:210-085-8
rtecs number:cb7250000
brn number:none
pubchem id:none
physical property data
physical property data:
1. character: yellow needle or flake crystal
2. melting point (℃):306℃
3. solubility:soluble in ethanol, ether and hot acetic acid. insoluble in cold water. soluble in ammonium hydroxide and alkali to form a reddish-yellow liquid. soluble in concentrated sulfuric acid to form a reddish-brown liquid
toxicological data
none yet
ecological data
3. ecological data:
1. other harmful effects: this substance may be harmful to the environment and should be harmful to water bodies. give special attention.
molecular structure data
5. molecular property data:
1, molar refractive index:60.55
2, molar volume (m3/mol):157.4
3, isotonic specific volume (90.2k):450.0
4, surface tension (dyne/cm):66.6
5, polarizability (10-24cm3 ):24.00
compute chemical data
1. reference value for hydrophobic parameter calculation (xlogp): none
2. number of hydrogen bond donors: 1
3. number of hydrogen bond acceptors: 3
4. number of rotatable chemical bonds: 0
5. number of tautomers: 5
6. topological molecule polar surface area 54.4
7. number of heavy atoms: 17
8. surface charge: 0
9. complexity: 349
10. number of isotope atoms: 0
11. determine the number of atomic stereocenters: 0
12. uncertain number of atomic stereocenters: 0
13. determine the number of chemical bond stereocenters: 0
14. number of uncertain chemical bond stereocenters: 0
15. number of covalent bond units: 1
properties and stability
1. basic properties
sublimable. can produce water-soluble barium salts.
storage method
storage:
seal the secret container and store it in a sealed main container in a cool place dry position.
synthesis method
2. introduction to production methods
mainly composed of anthraquinone-2-sulfonate or2-chloranthraquinone is obtained by hydrolysis. it can also be obtained from the roots of the plant umbelliferae.
purpose
3. purpose
for manufacturing2-methoxy,2-ethoxy,2-phenoxyanthraquinone or alizarin and other intermediates, dyes and other fine chemical products.
