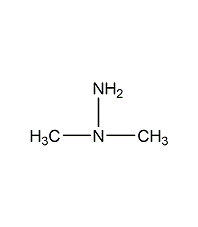
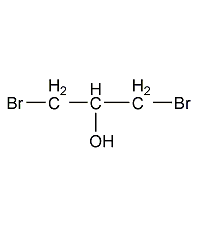
1,2-epoxypropane 1,2-epoxypropane
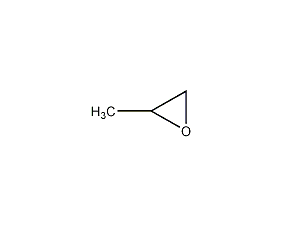

structural formula
| business number | 01jr |
|---|---|
| molecular formula | c3h6o |
| molecular weight | 58.08 |
| label |
propylene oxide, epoxy propylene, propylene oxide, propylene oxide, methyl ethylene oxide, propylene oxide, aliphatic alcohols, ethers and their derivatives |
numbering system
cas number:75-56-9
mdl number:mfcd00005126
einecs number:200-879-2
rtecs number:tz2975000
brn number:79763
pubchem number:24880314
physical property data
1. properties: colorless liquid with an ether-like odor. [1]
2. melting point (℃): -112[2]
3. boiling point (℃): 34[3]
4. relative density (water = 1): 0.83[4]
5. relative vapor density (air=1): 2.0[5]
6. saturated vapor pressure (kpa): 71.7 (25℃)[6]
7. heat of combustion (kj/mol): -1755.8[7]
8. critical temperature (℃): 209.1[8]
9. critical pressure (mpa): 4.93[9]
10. octanol/water partition coefficient: 0.03 [10]
11. flash point (℃): -37 (cc); -28.8 (oc) [11]
12. ignition temperature (℃): 449[12]
13. explosion upper limit (%): 36.0[13]
14. lower explosion limit (%): 2.3[14]
15. solubility: soluble in water, miscible in methanol, ether, acetone, benzene, tetracycline most organic solvents such as carbon chloride. [15]
16. viscosity (mpa·s, 0ºc): 0.410
17. viscosity (mpa·s, 20ºc): 0.327
18. heat of fusion (kj/mol): 6.5
19. specific heat capacity (kj/(kg·k), 15ºc, constant pressure): 1.95
20 .body expansion coefficient (k-1, liquid): 0.00213
21. combustion range in air (ml/100ml): 2.1~2.15
22 .critical density (g·cm-3): 0.305
23. critical volume (cm3·mol-1): 190
24. critical compression factor: 0.245
25. eccentricity factor: 0.271
26. lennard-jones parameter (a): 4.8515
27. lennard-jones parameter (k): 239.00
28. solubility parameter (j·cm-3)0.5: 19.110
29. van der waals area (cm2·mol-1): 4.640×109
30. van der waals volume (cm3·mol-1): 34.400
31. gas phase standard heat of combustion (enthalpy) (kj·mol-1): -1943.34
32. the gas phase standard claims heat (enthalpy) (kj·mol-1) : -94.68
33. gas phase standard entropy (j·mol-1·k-1): 281.15
34 .gas phase standard free energy of formation (kj·mol-1): -25.1
35. gas phase standard hot melt (j·mol-1· k-1): 72.55
36. liquid phase standard combustion heat (enthalpy) (kj·mol-1): -1915.44
37. liquid phase standard claims heat (enthalpythe conditions are reaction temperature 121°c and pressure 4.1 mpa. it was carried out in the presence of molybdenum catalyst, the reaction time was 0.5 h, the yield of propylene oxide was 88% (calculated as peroxide), and the selectivity was 81%. this method can co-produce tert-butyl alcohol with a yield of about 60%.
purpose
1. propylene oxide is an important organic chemical raw material and the third largest product of the propylene series. its largest use is to make polyether polyols and then polyurethane. in the distribution of uses in the united states and western europe, this use accounting for more than 60% and 70% respectively. used in the manufacture of nonionic surfactants and propylene alcohol, propylene glycol, alcohol ethers, propylene carbonate, isopropanolamine, propionaldehyde, synthetic glycerin, organic acids, synthetic resins, foam plastics, plasticizers, emulsifiers, and wetting agents , detergents, bactericides, fumigants, etc. fine chemicals derived from propylene oxide are used in almost all industrial sectors and in daily life. in addition, propylene oxide is also used in small amounts in coatings, brake fluids, antifreeze, jet engine fuel additives, floor polishes, printing inks, electronic chemicals, cleaners, mineral processing agents, leather processing, photosensitive fluids for ps plates, short-acting plasticizers, dyes, non-ionic surfactants, oil field demulsifiers, flame retardants, pesticide emulsifiers and wetting agents and other industries. also used in organic synthesis. it is used as a solvent for nitrocellulose, cellulose acetate, and various resins, a stabilizer for vinyl chloride resin and chlorine-containing solvents, and a fading inhibitor for nitrocellulose spray paint. it is also used in the manufacture of surfactants (wetting agents, detergents, emulsifiers, etc.) as well as medicines, pesticides, spices, and artificial leather.
2. used to prepare modified epoxy resin curing agent, synthetic resin and reactive diluent as epoxy resin adhesive. it is also used in the production of propylene glycol, propylene alcohol, propionaldehyde, polyether, isopropanolamine, higher fatty acid ester surfactants, plasticizers, medicines, pesticides, spices, and foam products. it is also a solvent for various resins, cellulose acetate, nitrocellulose, etc., a stabilizer for vinyl chloride resin and chlorine-containing solvents, a fading preventer for nitro spray paint, and can also be used as a bactericide, fumigant, wetting agent, etc. it is a broad-spectrum disinfectant that can kill bacterial propagules, spores, fungi and viruses. the general sterilization concentration is 800~2000mg/l.
3. it is an important raw material for organic synthesis. it is used to synthesize lubricants, surfactants, detergents, manufacture pesticides, and produce polyurethane foams and resins. [31]