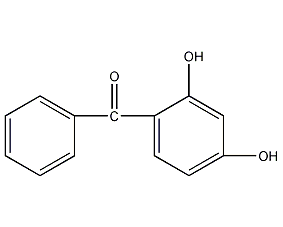
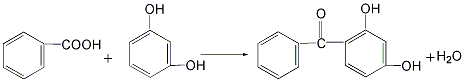2,4-dihydroxybenzophenone
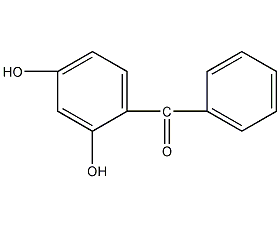

structural formula
| business number | 068z |
|---|---|
| molecular formula | c13h10o3 |
| molecular weight | 214.22 |
| label |
none yet |
numbering system
cas number:606-12-2
mdl number:none
einecs number:none
rtecs number:none
brn number:none
pubchem id:none
physical property data
physical property data:
1. character: light yellow needle crystal or white powder.
2. melting point (℃):142.6-144.6
3. boiling point (ºc,133.32pa):194
4. solubility: soluble in ethanol, ether, cold benzene and glacial acetic acid, virtually insoluble in cold water.
5. toxicity: orally administered to white ratsld50grey8600mg/kilogram, for mice2336mg/kilograms.
toxicological data
none
ecological data
3. ecological data:
1. other harmful effects: this substance may be harmful to the environment and should be harmful to water bodies. give special attention.
molecular structure data
5. molecular property data:
1, molar refractive index:59.80
2, molar volume (m3/mol):164.4
3, isotonic specific volume (90.2k ):456.9
4, surface tension (dyne/ cm):59.6
5, polarizability (10-24cm3): 23.71
compute chemical data
1. reference value for hydrophobic parameter calculation (xlogp): none
2. number of hydrogen bond donors: 2
3. number of hydrogen bond acceptors: 3
4. number of rotatable chemical bonds: 2
5. number of tautomers: 19
6. topological molecule polar surface area 57.5
7. number of heavy atoms: 16
8. surface charge: 0
9. complexity: 244
10. number of isotope atoms: 0
11. determine the number of atomic stereocenters: 0
12. uncertain number of atomic stereocenters: 0
13. determine the number of chemical bond stereocenters: 0
14. number of uncertain chemical bond stereocenters: 0
15. number of covalent bond units: 1
properties and stability
1. basic properties
irritant to eyes, respiratory system and skin.
storage method
2. storage
this product should be sealed and stored in a dark place.
synthesis method
3. brief description of production method
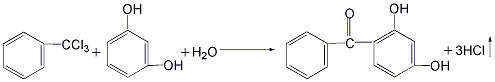
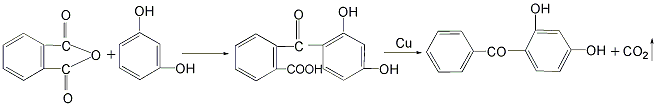
there are four main methods.1.resorcinol and benzoyl chloride as raw materials.2. uses resorcinol and trichlorotoluene as raw materials.3.made with benzoic acid and resorcinol as raw materials4.phthalic anhydride and resorcinol prepared by condensation2- ( 2′,4′-dihydroxybenzoyl)benzoic acid, obtained by dehydroxylation reaction2,4-dihydroxydi benzophenone.
purpose
4. purpose
used as uv absorber, derived from this product2-hydroxy-4-methoxybenzophenone (uv-9), 2-hydroxy-4-n-octyloxybenzophenone (uv-531) and other benzophenones, such compounds as well as other monohydroxy, dihydroxy, trihydroxy, and tetrahydroxy compounds are the most widely used absorption light stabilizers. can absorb290-400nano ultraviolet light, and has good compatibility with most synthetic resins capacitive, widely used in polymer materials. 2,4-dihydroxybenzophenone is suitable for polyvinyl chloride, polystyrene, epoxy resin, cellulose resin, unsaturated polyester, coatings and synthetic rubber, etc. maximum absorption wavelength range280-340nano, general usage0.1-1%. however, the light stabilizing effect of this product is not outstanding.
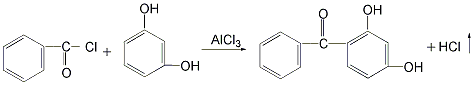 2. use resorcinol and trichlorotoluene as raw materials in the reaction kettle add resorcinol and appropriate amount of water and stir to dissolve. then add trichlorotoluene and an appropriate amount of alcohol, and stir to make the materials completely miscible. the reaction was carried out at 40°c, and 2,4-dihydroxybenzophenone was generated and precipitated as a solid. after the reaction is completed, the reactants are filtered, and the filter cake is washed with dilute sodium bicarbonate solution and then dried to obtain the finished product. the yield of this method can reach 95%, the raw materials are easy to obtain, and the product cost is low. however, the product has a darker color and is not easy to decolorize and purify.
2. use resorcinol and trichlorotoluene as raw materials in the reaction kettle add resorcinol and appropriate amount of water and stir to dissolve. then add trichlorotoluene and an appropriate amount of alcohol, and stir to make the materials completely miscible. the reaction was carried out at 40°c, and 2,4-dihydroxybenzophenone was generated and precipitated as a solid. after the reaction is completed, the reactants are filtered, and the filter cake is washed with dilute sodium bicarbonate solution and then dried to obtain the finished product. the yield of this method can reach 95%, the raw materials are easy to obtain, and the product cost is low. however, the product has a darker color and is not easy to decolorize and purify.