6-methoxy-8-nitroquinoline 6-methoxy-8-nitroquinoline
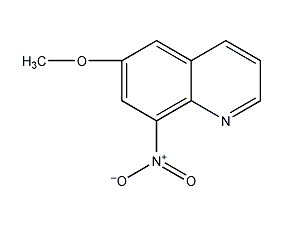

structural formula
| business number | 01w9 |
|---|---|
| molecular formula | c10h8n2o3 |
| molecular weight | 204.18 |
| label |
none yet |
numbering system
cas number:85-81-4
mdl number:mfcd00006802
einecs number:201-633-7
rtecs number:vc0365000
brn number:none
pubchem number:24852410
physical property data
1. character: yellow needle crystal
2. density (g/ml ,25/4℃): undetermined
3. relative vapor density (g/ml,air=1): undetermined
4. melting point (ºc):159-160℃
5. boiling point (ºc,normal pressure): undetermined
6. boiling point (ºc,5.2 kpa): undetermined
7. refractive index: undetermined
8. flash point (ºc): undetermined
9. specific optical rotation (º ): undetermined
10. autoignition point or ignition temperature (ºc): undetermined
11. vapor pressure (kpa, 25ºc): undetermined
12. saturated vapor pressure (kpa ,60ºc): undetermined
3. isotonic specific volume (90.2k):417.4
4. surface tension (dyne/cm):55.9
5. polarizability(10-24cm3):21.96
compute chemical data
1. reference value for hydrophobic parameter calculation (xlogp): none
2. number of hydrogen bond donors: 0
3. number of hydrogen bond acceptors: 4
4. number of rotatable chemical bonds: 1
5. number of tautomers: none
6. topological molecule polar surface area 67.9
7. number of heavy atoms: 15
8. surface charge: 0
9. complexity: 241
10. number of isotope atoms: 0
11. determine the number of atomic stereocenters: 0
12. uncertain number of atomic stereocenters: 0
13. determine the number of chemical bond stereocenters: 0
14. number of uncertain chemical bond stereocenters: 0
15. number of covalent bond units: 1
properties and stability
none
storage method
sealed at4℃save in a dry place and away from light.
synthesis method
by4-methoxy-2-nitroaniline is obtained by cyclization reaction. add glycerol into the reaction pot and heat at 70℃add sulfuric acid below and then put in4-methoxy-2-nitroaniline. at75-80℃after insulating, add an aqueous solution of potassium iodide and iodine, and raise the temperature to 125℃, and keep warm until the reaction reaches the end point. dilute with ice water and incubate at 35℃the following is neutralized with sodium hydroxide solution ph the value is3.5, filter, wash to phvalue is 5, got6-methoxy-8-nitroquinoline. the yield is 90%.
purpose
this product can be used as an intermediate for the antimalarial drug primaquine phosphate.
y: arial; mso-hansi-font-family: arial; mso-bidi-font-family: arial”>add sulfuric acid below and then put in4-methoxy-2-nitroaniline .at75-80℃after insulating, add an aqueous solution of potassium iodide and iodine, and raise the temperature to 125℃, incubate until the reaction reaches the end point. dilute with ice water, in35℃neutralize with sodium hydroxide solution belowph the value is3.5, filter, wash to phvalue is56-methoxy-8-nitroquinoline. the yield is 90%.
purpose
this product can be used as an intermediate for the antimalarial drug primaquine phosphate.